What's Best For A Spa: Chlorine or Bromine?
Peruse any of the Internet forums devoted to pool and spa topics and you'll find lots of misinformation out there, particularly when it comes to water quality in spas. These "hot tubs" for the residential market are touted as oases of pleasure and a remedy for the aches of middle age—and they're all that!—however, buyers need to be properly informed about water quality maintenance too. Owing to their operating range of 96°F–104°F, jetted water, and a sanitizer demand that can be variable in the extreme, spas create an environment where microbes will flourish unless the owner is vigilant about sanitizing the water.
In the absence of adequate sanitizer, bacteria reproduce quickly. This can present a serious situation since some strains cause infections and others cause major gastrointestinal illnesses. For instance, the most common spa-related ailment, dermatitis, is caused by bacteria. Another bacteria, Legionella pneumophila, can even be fatal if inhaled with the mist from a spa. Viruses, protozoans, and algae will multiply rapidly in inadequately treated water, as will the biofilm that can shelter microorganisms.
Considering that an adult bather sheds about a billion bacteria when entering a spa, the first step in maintaining excellent water quality should be taking a soapy shower before entry. The second step is to continually destroy all microbial invaders through sanitation and any nonliving contaminants through oxidation. The third step is to keep the filter clean and run the filtration system for the manufacturer's recommended time each day so all the water is adequately treated.
Spa owners need to be well informed about the importance of testing and treating their water on a regular basis.
"But My Spa's Ozonated"
Most spas sold today come already equipped with some type of ozone system. As they pass by the "ozonator," both microscopic organisms and nonliving impurities are destroyed. Unfortunately, once out in the main body of water, ozone dissipates too quickly to provide adequate protection against germs. Optimally, ozone's half-life is about 20 minutes, but in the real world it's gone much quicker as the bather load increases. Therefore, even if a spa is equipped with an ozonator, a supplemental sanitizer must be employed that will leave a residual for ongoing protection of bathers.
Due to simply a difference in atomic weight, for a DPD test, bromine is roughly twice the concentration as chlorine.
The Dynamic Duo
The most popular sanitizers on the market today are chlorine and bromine, which are both classified as halogens. Halogens are highly reactive elements, a characteristic that makes them ideal for sanitizing and oxidizing contaminants in water. Chlorine is slightly more reactive than bromine, making it somewhat more effective. The other reason why the ideal range for residual bromine is a little higher than for residual chlorine is that, owing to their relative atomic weights, it takes roughly twice as much bromine, as ppm, to get the same oxidizing capacity as chlorine. See Table 1.
If a spa has an ozonator, the recommended level of residual disinfectant remains the same; however, the amount of chlorine or bromine product needed to achieve this goal will be less because the ozone generated is helping to sanitize and oxidize the water.
In their elemental forms and under standard conditions for temperature and pressure, chlorine exists as a pale green gas, bromine as a reddish-brown liquid. These are dangerous and not used to treat spas. However, certain formulations have approval from the U.S. Environmental Protection Agency for spa use. How do you choose which product is best to use? There is a dizzying array of brands on the market, so for this survey we will consider broad categories and use typical figures for our stats as we explore the advantages and disadvantages of each. But first, an important point made by the manufacturers: Proper treatment of spa water is easiest when a program approach is followed. Keeping with a program ensures that the products used for sanitation, oxidation, and water balance work well together.
|
Table 1: Guidelines for Sanitizers in Spas |
|||
|---|---|---|---|
| Sanitizer | Minimum | Ideal Range | Maximum |
| Free chlorine, ppm | 2.0 | 3.0–5.0 | 10.0 |
| Total bromine, ppm | 2.0 | 4.0–6.0 | 10.0 |
Chlorine Choices
Among the five general types of chlorine products sold for use with recreational water, lithium hypochlorite (a fast-dissolving granule) is the most costly of the group. However, it's the safest to handle and can be stored for a long period without losing its potency. When a spa cover is off, UV rays from sunlight will quickly deplete any form of chlorine that hasn’t been "stabilized" with cyanuric acid (CYA), including lithium hypo. One ppm of unstabilized chlorine will dissipate in 15 minutes or less under direct sunlight. With heated spa water, this dissipation rate is even faster. The loss can be controlled with a separate, one-time addition of CYA. That way the stabilizer won't build up unintentionally, taking you over the recommended level of 30–50 ppm.
Unlike its cousin calcium hypochlorite, lithium hypo doesn't add calcium hardness to water, but it will raise the total dissolved solids level more than other products compared here. It's the most expensive form of chlorine and yet it only provides 35 percent available chlorine content (ACC), so comparatively you use more. Because it's very alkaline, having a pH around 11 will drive pH up and most likely total alkalinity too; dry acid must be added periodically to counter this.
The effect any hypochlorite-based chlorine has on pH, and therefore on water balance in general, is the main reason why spa treatments with dichlor (the chlorinated isocyanurate, sodium dichloro-s-triazinetrione) sell much better. Dichlor is almost pH neutral, being about pH 6.7. Another granular product that’s usually broadcast on the water's surface, dichlor dissolves rapidly but not as fast as lithium hypochlorite. It has a good shelf life, and the anhydrous form provides 63 percent ACC, the dihydrate form 56 percent ACC. On the downside, this stabilized chlorine contributes heavily to the buildup of cyanuric acid. In a 500-gallon spa, a recommended maintenance dose of the dihydrate form of one-half ounce will add over 4 ppm of stabilizer to the water. For every pound of dichlor added, 5 ppm of cyanuric acid (CYA) is also added for every 10,000 gallons of water. Do that several times a week and it’s not long before the limit for CYA is exceeded. Shocking with dichlor accelerates the buildup. The only recourse is to partially drain the spa and refill with fresh water.
For this reason, some people will switch over to using pool-grade bleach (sodium hypochlorite). Do NOT use laundry-grade bleach as it is weaker in strength and will contribute greatly to TDS levels. Sodium hypochlorite is very alkaline (pH of 11–13). The total dissolved solids from sodium hypochlorite, plus the total dissolved solids contributed by the acid used to counter its high pH, mean an ever-increasing TDS level; however, spa water should be replaced often enough so TDS buildup does not become an issue.
What is of more concern is sodium hypochlorite's ACC of only 10–12 percent which naturally degrades over time. Also, application is trickier. You can easily bleach the spa and your clothes as well as the water. Finally, if the choice is to alternate between stabilized and unstabilized chorine, they should not be stored near each other. The reaction of an isocyanurate with hypochlorite can result in rapid formation of nitrogen trichloride, which at high enough concentrations will combust spontaneously with great violence.
Generally the best bargain pricewise, calcium hypochlorite (aka cal hypo) products can have a high pH, from 8.5 to over 11. Watch the water's pH and total alkalinity carefully, adding liquid or dry acid as needed. Applying cal hypo is not quite as easy as lithium hypo or dichlor. Premixing granular cal hypo in a bucket of water before adding it to the spa is recommended to prevent discoloration caused by the slow dissolving granules. Spa supplies dealers and homeowners alike must take great care in handling and storing cal hypo products to prevent a fire. In recent years some manufacturers have been reformulating their blends to lower the ACC and therefore lessen the hazard. Consumers now can see products with an ACC ranging from 45–78 percent.
Trichloro-s-triazinetrione, or simply trichlor, is another sanitizer and stabilizer all in one. Trichlor tablets or sticks with 90 percent ACC may be used in large built-in commercial spas with chlorinators…but with caution, as its acidic nature (pH 2.8–3.5) lowers pH and total alkalinity, wreaking havoc with the heater elements and metal pipes, fittings, and surfaces unless increased with soda ash or sodium bicarbonate. The erosion of trichlor is accelerated by warm water, making it easy to accidently overdose a spa. For these reasons, using trichlor tabs or a granular trichlor shock/algae treatment in a spa is not advised and may void the spa’s warranty.
Besides these off-the-shelf products, generators that use salt to produce chlorine can be employed to chlorinate the water in spas.
Chlorine Residuals
Once added to water, any type of chlorine will form hypochlorous acid (HOCl). Hypochlorous acid is constantly dissociating, i.e., breaking up into hypochlorite ions (OCl-) and hydrogen ions (H+) and then reforming into hypochlorous acid again. Together HOCl and OCl- make up free chlorine, the chemical species that destroys microorganisms and oxidizes organics. However, HOCl is a much more potent disinfectant than OCl-. Free chlorine’s effectiveness as a sanitizer/oxidizer is solely pH-dependent. At the lower end of the acceptable pH range of 7.2, nearly 67% of free chlorine is in the form of HOCl. At the 7.8 level, it drops to almost 33%. At a pH of 7.5, HOCl is 50%.
When free chlorine reacts with sweat, urine, and other ammonia and organic-nitrogen compounds introduced to the spa, it forms foul-smelling, eye- and throat-irritating compounds known as chloramines or combined chlorine. Chloramines are noticeable to bathers at levels as low as 0.2 ppm and must be eliminated by superchlorinating to "breakpoint" when they reach 0.5 ppm or sooner. If the spa is equipped with an ozonator, chloramines will be destroyed while it's running. Note: Potassium monopersulfate shocks are not very effective at eliminating chloramines but do help prevent their formation.
Testing Chlorine
Free and combined chlorine levels may be monitored using DPD and FAS-DPD test methods and some test strips but not with the OT method. OT cannot distinguish between free and combined chlorine, making it an inferior test method for spa care. While slightly more expensive and a little more time-consuming, an FAS-DPD titration has several advantages over the DPD colorimetric method.
First, with FAS-DPD there's no need to find a color match between the pink of the treated sample and a set of pinkish color standards. People who are colorblind for red—about 6–8 percent of men and some women—particularly find this of great benefit. Second, the pink indicator in the FAS-DPD test won't bleach out around 10 ppm of chlorine like it does with DPD; there's less chance a tester will think he has no chlorine in the water when he really has a lot. FAS-DPD also will detect those pesky chloramines as low as 0.2 ppm. With the most commonly used color comparators, the best a DPD test can do is 0.5 ppm. You can accurately read free chlorine up to 20 ppm with FAS-DPD using a 10 mL sample (1 drop = 0.5 ppm), whereas a DPD color comparator or test strip only works up to 10 ppm. Therefore, FAS-DPD is the better choice for monitoring superchlorinated (shocked) water to know when it is safe to reenter.
Spa owners are best advised to use a combination test kit with an FAS-DPD test in it for monitoring free and combined chlorine levels, such as the K-2006. Consumers who prefer the convenience of test strips for routine testing can augment their arsenal with our stand-alone FAS-DPD test, K-1515-A, for troubleshooting.
K-2006 (left) and K-1515-A (right)
A Quick Review of Bromine
Consider these facts about bromine:
- It's an effective sanitizer (i.e., destroys microorganisms, such as bacteria and algae, to an acceptable level that will not cause harm to bathers), but…
- Compared to chlorine, ozone, and potassium monopersulfate, it's weaker when it comes to oxidizing organic compounds (i.e., scouring nonliving contaminants out of the water, such as bather wastes, personal care products, pollen, and dust).
- Elemental bromine (Br2) exists as a reddish-brown liquid and is too dangerous to use as a spa treatment.
- To keep bathers healthy, the bromine level should not dip below 2.0 ppm.

Before entering a spa, the water should always be tested to assure there is an adequate level of sanitizer.
Advantages to Using Bromine
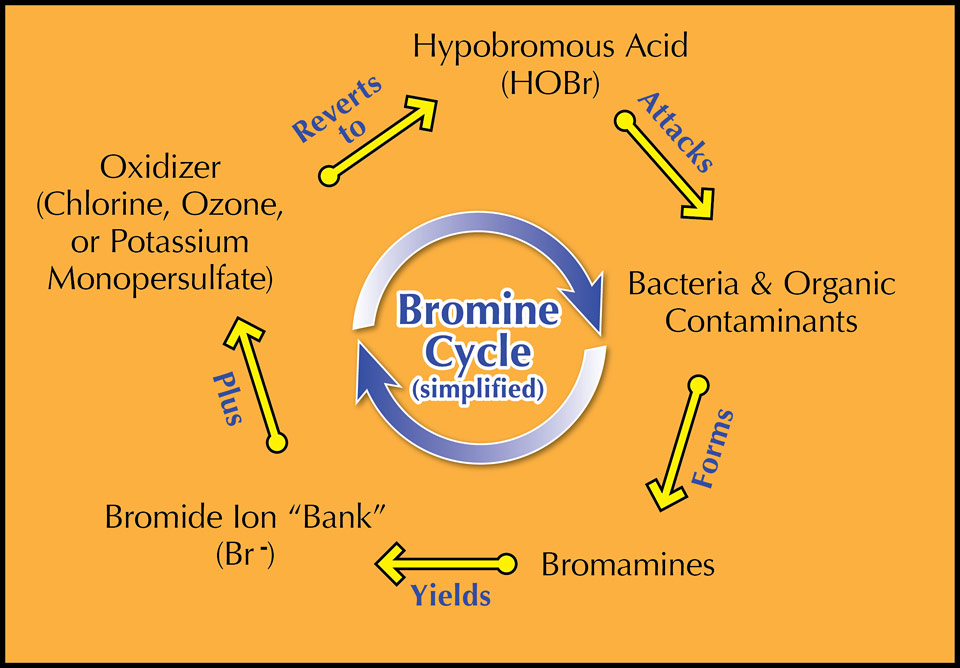
Bromine is easy to use. There are two main ways to dose bromine manually. One way is to establish what's called a bromide bank by putting a threshold quantity (15–30 ppm) of a harmless bromide salt into the water. You then introduce an oxidizer, sometimes labeled "activator," to convert these bromide ions to a form that will kill microorganisms. The oxidizer/activator could be potassium monopersulfate—also listed on products as potassium peroxymonosulfate; it's the active ingredient in most non-chlorine shock treatments—or chlorine. To many, learning that chlorine can have a place in a bromine-sanitized spa comes as a surprise. The second way is to apply a hydantoin product that already contains oxidized bromine, using a specially-sized floater or feeder. The oxidized bromine is gradually released as the tablets react with the water. Both methods result in the formation of hypobromous acid, HOBr, and hypobromite ions, OBr-. A third way to produce HOBr and OBr- is through electrolytic conversion of the bromide salt with an automatic bromine generator.
In this duo, hypobromous acid is the champion pollutant fighter. It is not nearly as particular about pH as its chlorine counterpart, hypochlorous acid. At pH 6, nearly 100% of bromine is in the more reactive HOBr form; at the same pH, 97% of free chlorine would be in the form of HOCl. But at pH 8, while 83% of active bromine is present as HOBr, only 24% of free chlorine would be in its more reactive hypochlorous acid state at any given moment. Since pH can fluctuate dramatically in a spa, having a sanitizer that's able to work over a wide pH range is an important asset.
Spas promote heavy sweating, and urine often is involuntarily leaked into the water as well. It has been reported that the average person produces about one pint of sweat during a 15-minute soak in a spa. The bromamines formed in reaction with ammonia-laden bather wastes don't have the unpleasant odor of chloramines and they are less irritating to the eyes and respiratory tract. Moreover, bromamines actively disinfect the water until they self-destruct a short time after their formation. Still, because bromine is a weaker oxidizer, periodic shocking with chlorine or potassium monopersulfate is strongly advised no matter what program is chosen. The Association of Pool & Spa Professionals (APSP) says supplemental oxidation is particularly important when using bromine compounds to prevent chemical rashes. Shocking will eliminate contaminants bromine cannot, as well as regenerate the bromide bank.
Once on Bromine, Always on Bromine
What’s amazing about bromine chemistry is when hypobromous acid has done its job, most of it reverts to bromide ions. The sanitizing cycle will begin anew just as soon as the ions in the bank meet up with an oxidizer! (See graphic.) Remember, chlorine will sacrifice itself to making HOBr as long as there’s a bromide level of 15 ppm or more. This means once you start a bromine program you can't switch to a chlorine program unless those bromide ions are removed first.
What's Not to Like?
The drawbacks to using bromine sanitizers are:
- Tendency to deplete total alkalinity; corrosion damage to the heater can result if testing is sporadic and unbalanced water is not detected and treated
- No effective protection from degradation by sunlight comparable to cyanuric acid's effect on chlorine (mitigated by the fact most spas are covered a lot of the time)
- Program cost may be higher than chlorine
- No test for just the bromide ion level
- The impossibility of switching from bromine to chlorine without draining the spa
But bromine's advantages outweigh all of these for a great many spa owners.
Bromine Choices
For many years bromine sanitation of spas has been accomplished with a bromide salt in either liquid or granular form (such as sodium bromide, which has a pH of 6.5 to 8), coupled with a separate granular oxidizer ("activator"), usually potassium monopersulfate specially buffered for use in spas to neutralize its acidity. Generally, usage instructions for this two-step system say to manually add sufficient product to establish a 30 ppm bromide reserve each time the spa is filled. A smaller maintenance dose may be recommended sometime thereafter or following heavy use. It's important to realize sodium bromide itself is not a disinfectant. It must be used in conjunction with the activator, which is added periodically to convert the bromide bank to the killing form of bromine. With this system, no floater or feeder is necessary.*
A newer formulation is BCDMH + DCDMH + DCEMH (1-bromo-3-chloro-5,5-dimethylhydantoin + 1,3-dichloro-5,5-dimethylhydantoin + 1,3-dichloro-5-ethyl-5-methylhydantoin), sometimes referred to as DantobromTM S. In the spa market, this is sold in both tablet and briquette form. The compound has a pH of 3.6 and an equivalent available chlorine content of 62 percent. In spas it can be dispensed in a simple floater (if the warranty permits this) or placed in an erosion-soaker feeder. It's necessary to add sodium bromide to build a bromide reserve when this product is used for the first time and whenever the water is replaced. This bromine treatment is acidic so pH and alkalinity should be monitored closely.
A chlorine-free hydantoin approach is DBDMH (1,3-dibromo-5,5-dimethylhydantoin). It's sold as slow-dissolving nuggets or tablets; in a spa either can be applied using an approved feeder or floater. Shocking regularly with any conventional treatment except hydrogen peroxide is advised. DBDMH disinfectants have a nearly neutral pH, e.g., 6.6; an equivalent available chlorine content as high as 54 percent; and good shelf life when stored in a cool, dry place.
Testing Bromine
Because bromamines do not have the objectionable properties of chloramines, bromine sanitizer tests do not need to differentiate between the free and combined forms. The total bromine residual can be read with OT, DPD, FAS-DPD, and some test strips. Perform the test immediately after taking the water sample to get the most accurate reading.
All Taylor Residential™ liquid kits test for total bromine and either free or total chlorine, making them handy for homeowners who prefer chlorine for their pools but bromine for their spas. K-1005 shown.
Authorities say 4.0–6.0 ppm is the ideal concentration of bromine in spas. Opinions differ on the maximum allowable while soaking. The National Swimming Pool Foundation® says 10.0 ppm, whereas ANSI/APSP does not cite any recommended maximum level. Note: Since cyanuric acid stabilizer doesn't work with bromine, there is no need for a CYA test in kits that only contain a bromine sanitizer test, such as Taylor's Complete™ FAS-DPD kit (K-2106).
Conclusion
So, which is the best choice for maintaining the water quality in a spa? The answer is not clear-cut. In the end, it comes down to how much use the spa gets; one's understanding of hot-water chemistry, particularly how each of the factors in water balance affects the others; how much time can be devoted to testing and treatment; what one is willing to spend; and even whether water restrictions prevent frequent dumping of the spa. But with the information in this article, spa owners and the industry members who serve them can make an informed choice.
* The concept of equivalent available chlorine content does not apply here, as the level of bromine released is not dependent upon the two chemicals but on the ratio of their application in a given volume of water.