Testing Alkalinity In Boiler Water
Alkalinity is a key control parameter for boilers (outside of the prescribed operating range, it can lead to corrosion, caustic metal embrittlement, and carryover) and cooling water systems (where it has a significant effect on chemical treatments for scale and corrosion inhibition).
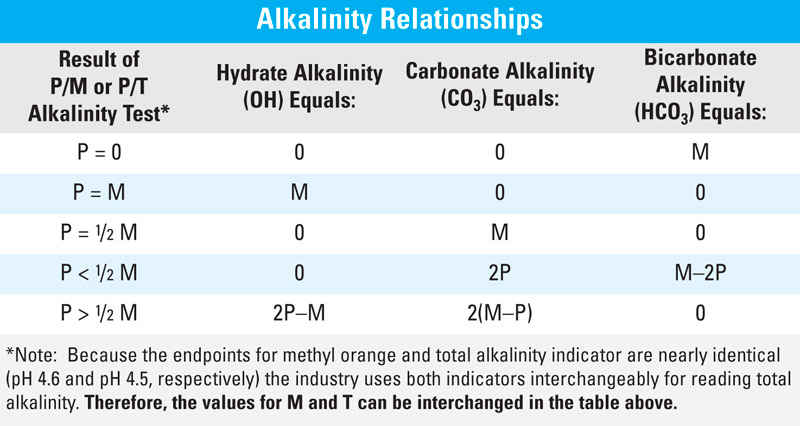
P, M, and T
Depending upon the pH of the water, alkalinity can exist in water in three basic forms: carbonate (CO3), bicarbonate (HCO3), or hydroxide (OH). Total alkalinity is the sum of these three forms. Alkalinity is measured by titrating a water sample with a standard acid to a designated pH and is recorded as P, M, or T alkalinity. P alkalinity is titrated with phenolphthalein to pH 8.3, M alkalinity with methyl orange indicator to pH 4.6, and T alkalinity with total alkalinity indicator to pH 4.5. Once you know the values for P, M, or T alkalinity you can use the table of relationships below to determine treatment control and effectiveness.
Alkalinity Relationships
In boiler water, treatment specialists must maintain M (or T) alkalinity below a maximum level prescribed by the boiler's manufacturer to help prevent foaming and carryover. Typically, as boiler operating pressure increases so does the need for higher purity water. At the same time, water treaters need to maintain a minimum level of OH alkalinity (generally, over 150 ppm as CaCO3) to prevent scaling tendencies while ensuring excess alkalinity doesn't interfere with other chemical treatment components. In cooling water, treatment professionals aim to control alkalinity (and pH) to prevent corrosion and scaling. Below acceptable levels, these parameters can contribute to corrosion. The higher the pH, the more alkalinity is present in the carbonate species, which can react with calcium in the water to form scale.
Test Interferences
There are some interferences you should be aware of when performing alkalinity titrations. Highly turbid or colored samples may mask the color change at the endpoint. The suspended solids can be removed by standard paper filtration methods, while colloidal color may require the use of syringe filtration (6247, 6249; 6248, 6257, 6261). High levels of chlorine may interfere with the indicators. Add one drop of sodium thiosulfate to prevent this. Treatment polymers will titrate as M and T alkalinity. In waters using polymer treatment, OH alkalinity needs to be directly titrated using the barium chloride method. Using a 2P-M method to determine OH alkalinity will result in a negative interference for the calculation of OH. Absorption of CO2 will depress alkalinity in water samples. To prevent this situation, analyze samples at the point of collection. When on-site analysis is not practical, collect samples for off-site testing in overflowing containers and cap them tightly. If acid is being added to adjust pH, it will also alter the P/T alkalinity relationship.